Capsule filling machine manual kicks off with a comprehensive overview of the equipment’s purpose, function, and various types available, including semi-automatic and automatic machines. This opening paragraph sets the stage for a detailed discussion on the machine’s components, operations, safety features, and regulatory compliance. With a brief history of capsule filling machine development, we delve into the intricacies of the equipment, ensuring readers grasp its significance in the manufacturing process.
The capsule filling machine manual is an essential guide for operators, maintenance personnel, and quality control teams to understand the machine’s functionality, ensure its proper maintenance, and adhere to regulatory standards. By following this manual, readers will gain a solid understanding of the machine’s components, operations, and troubleshooting procedures, enabling them to optimize production efficiency and product quality.
Introduction to Capsule Filling Machines
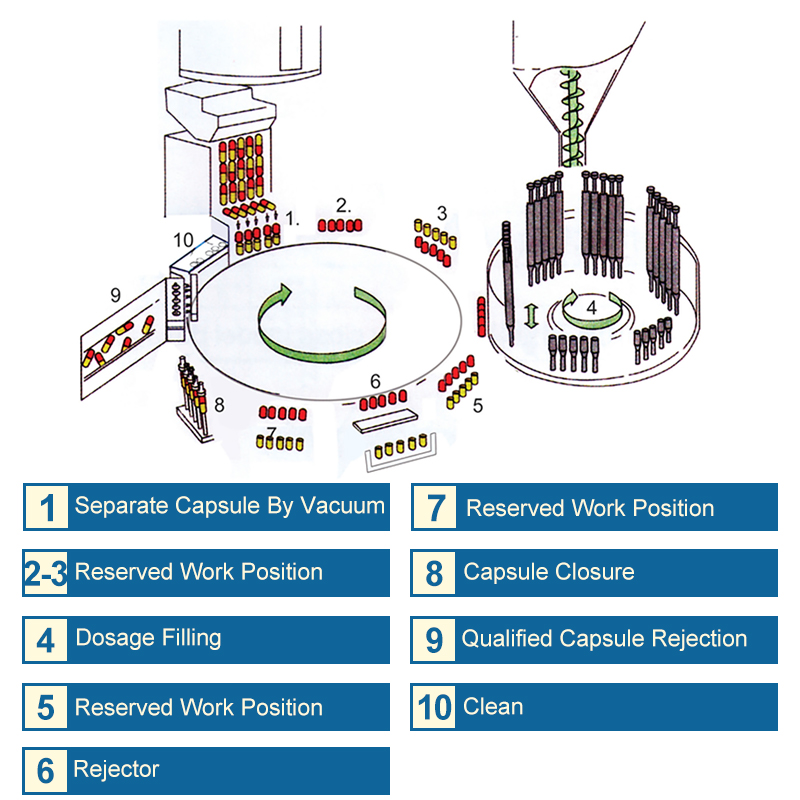
Capsule filling machines play a crucial role in the pharmaceutical industry by automating the process of filling capsules with powdered or liquid medication. These machines are designed to meet the increasing demand for precision, speed, and efficiency in pharmaceutical manufacturing.
Purpose and Function of a Capsule Filling Machine
A capsule filling machine is a device that automatically fills capsules with the desired amount of powdered or liquid medication. Its primary function is to ensure accurate and consistent dosing, while also minimizing waste and optimizing production time. The machine typically consists of a hopper for storing the medication, a filling mechanism for accurately measuring the dosing, and an ejection system for disposing of empty capsules or partially filled capsules.
Types of Capsule Filling Machines
There are two main types of capsule filling machines available: semi-automatic and automatic.
Semi-Automatic Capsule Filling Machines
Semi-automatic machines require manual intervention at various stages of the filling process. They are ideal for small-scale production, pilot productions, or for pharmaceutical companies that require flexibility in their production settings.
Automatic Capsule Filling Machines
Automatic machines are designed for high-speed production and are typically used in large-scale pharmaceutical manufacturing. They offer improved precision, increased efficiency, and reduced labor costs.
Brief History of Capsule Filling Machine Development
The first capsule filling machines were introduced in the mid-20th century, with early machines consisting of manual, labor-intensive processes. Over the years, advancements in technology have led to the development of semi-automatic and automatic machines, which have improved the precision, speed, and efficiency of capsule filling processes.
Early Development (1940s-1960s)
The first capsule filling machines were manual machines that relied on workers to fill capsules by hand. These machines lacked precision and were time-consuming.
Mechanical Advancements (1970s-1990s)
The introduction of mechanical filling machines marked a significant improvement in the precision and speed of capsule filling. These machines could fill capsules in a matter of seconds, but still required manual intervention at various stages.
Computerization and Automation (2000s-Present)
The advent of computerization and automation has led to the development of high-speed, precise, and efficient capsule filling machines. These machines can fill capsules at rates of up to 100 capsules per minute and offer improved accuracy through advanced sensors and control systems.
Machine Components and Operations
The capsule filling machine is composed of several key components that work together to fill, seal, and eject capsules. Understanding the functionality of these components is crucial for operating the machine efficiently and effectively.
Primary Machine Components
The capsule filling machine consists of three primary components: dosers, fillers, and sealers. These components work in conjunction to fill and seal capsules with precise amounts of powder or liquid.
The doser is responsible for measuring and dispensing the required amount of powder or liquid into the capsule. It is equipped with a precision metering system that ensures accurate and consistent dosing. The doser is typically designed with interchangeable containers to accommodate different types and quantities of materials.
The filler is responsible for filling the capsule with the measured amount of powder or liquid dispensed by the doser. It is designed to ensure precise filling accuracy, and some models may include features such as automatic capping and filling height adjustment.
The sealer is responsible for sealing the capsule after it has been filled. It is typically equipped with a heating element that applies heat to the capsule to ensure a tight seal. Some models may also include features such as automatic ejection and capsule counting.
Filling, Sealing, and Ejecting Capsules
The capsule filling machine operates on a continuous cycle, with each step carefully synchronized to ensure efficient and accurate processing. Here is a brief overview of the filling, sealing, and ejecting process:
1. Dosing: The doser measures and dispenses the required amount of powder or liquid into the capsule.
2. Filling: The filler fills the capsule with the measured amount of powder or liquid dispensed by the doser.
3. Sealing: The sealer applies heat to the capsule to create a tight seal.
4. Ejection: The sealed capsule is ejected from the machine and collected in a container.
Machine’s Control Panel and User Interface
The machine’s control panel is designed to be user-friendly and intuitive, allowing operators to easily navigate and adjust settings as needed. The user interface typically includes a range of features, such as:
* Display Screen: A display screen that shows real-time information about the machine’s operation, including filling accuracy, sealing temperature, and ejection rate.
* Buttons and Controls: A set of buttons and controls that allow operators to adjust settings and navigate the menu.
* Sensor Indicators: A set of sensor indicators that monitor the machine’s performance and alert operators to any potential issues.
Error Messages and Solutions
The machine’s control panel is equipped with a range of error messages that alert operators to potential issues with the machine’s operation. Here are some common error messages and their solutions:
* Error Message 1: “Doser failure – check container and adjust settings”.
+ Solution: Check the doser container for damage or contamination and adjust the settings to ensure accurate dosing.
* Error Message 2: “Filler error – check filling accuracy”.
+ Solution: Check the filler’s accuracy by comparing the filled capsules to a reference standard.
* Error Message 3: “Sealer malfunction – check heating element”.
+ Solution: Inspect the heating element for damage or malfunction and replace it if necessary.
Capsule Specifications and Design

To function efficiently, a capsule filling machine requires accurate and precise capsule specifications. Ensuring proper capsule dimension consistency is crucial to avoid mechanical issues and contamination risks.
Determining Capsule Size and Material
Capsule size and material selection primarily depend on the type of medication being filled into the capsule. There are various capsule sizes available, ranging from 0.75 ml to 0.96 ml.
* To determine the ideal capsule size, consider the volume of the medication and space available in the capsule.
* The most common capsule materials are gelatin and vegetarian capsules made from plant-based materials such as potato starch or rice flour.
* Gelatin-based capsules are suitable for medications that are heat-sensitive or easily oxidized.
Ensuring Capsule Dimension Consistency
Proper capsule dimension consistency is essential for the machine’s performance and the quality of the filled capsules.
* Capsules of varying sizes can cause the machine’s precision mechanisms to malfunction, affecting the uniformity of the filled capsules.
* A single misplaced capsule can prevent the machine from working correctly, leading to delays and waste.
* Maintaining a consistent supply of capsules with the same size and material will ensure smooth operations.
Creating or Sourcing Custom Capsule Designs
Sometimes, a specific customized capsule design may be required to cater to a particular medication or customer request.
* Capsule manufacturers can create custom capsule designs based on the provided specifications and material requirements.
* Sourcing custom capsules from specialized manufacturers is also an option, especially for large-scale orders or short lead times.
* Ensure that the chosen manufacturer adheres to regulatory guidelines and quality standards.
Factors Affecting Capsule Weight and Quality
Capsule weight and quality directly impact the medication’s effectiveness and patient compliance.
* Capsule weight affects the dosage and stability of the medication; an incorrect capsule size can lead to medication inaccuracies.
* Material choice influences capsule stability and compatibility with the medication’s active ingredients.
* Manufacturing processes, such as temperature control, moisture levels, and cleanliness, also impact the quality of the capsule.
* Capsule filling machines should operate within the manufacturer’s guidelines to minimize the risk of contamination and maintain consistent quality.
Critical Dimensions and Capacities
Familiarize yourself with key capsule specifications and capacities:
| Dimension | Description | Standard Tolerances |
| — | — | — |
| Size (ml) | Volume capacity | ±5% |
| Material | Material thickness | ±20% |
| Length (mm) | Capsule length | ±1 mm |
| Diameter (mm) | Capsule diameter | ±0.2 mm |
| Weight Range | Gross weight | ±5% |
Regularly check and adjust your machine settings according to the specifications to ensure optimal performance.
Please refer to the manufacturer’s documentation for specific machine setup and calibration instructions.
Machine Calibration and Troubleshooting
Regular machine calibration and adjustments are crucial to ensure optimal performance, accuracy, and efficiency of the capsule filling machine. Over time, the machine’s mechanisms and sensors can wear and tear, leading to discrepancies in filling weights, capsule sealing, or machine operation. Therefore, periodic calibration is necessary to maintain the machine’s reliability and effectiveness.
Necessity of Regular Calibration
Machine calibration involves fine-tuning the machine’s parameters to ensure that it operates within the specified range. This process helps to:
– Achieve precise filling weights by adjusting the dosing mechanism to account for any deviations in the machine’s performance.
– Optimize capsule sealing by calibrating the sealing system to ensure tight seals and minimize defects.
– Ensure smooth machine operation by adjusting parameters such as motor speed, pressure, and timing to prevent vibrations, noise, or other issues.
– Improve overall efficiency by reducing waste, reducing downtime, and increasing production output.
Methods for Identifying and Solving Common Technical Issues
To identify and solve common technical issues, operators should follow a systematic approach:
– Regular Maintenance: Schedule regular maintenance tasks, such as cleaning, lubricating, and replacing worn-out parts, to prevent machine degradation.
– Performance Monitoring: Continuously monitor machine performance, checking for any deviations in filling weights, capsule sealing, or other parameters.
– Troubleshooting Guides: Refer to the machine’s troubleshooting guide or consult the manufacturer’s documentation to identify potential causes and solutions for common issues.
– Technical Support: Contact the manufacturer’s technical support team or a qualified technician for assistance with complex issues that cannot be resolved through troubleshooting.
Key Performance Indicators (KPIs) for Capsule Filling Machine Operation
To evaluate the machine’s performance and effectiveness, operators should track and analyze the following KPIs:
– Filling Accuracy: Measure the deviation in filling weights to ensure that they fall within the specified range.
– Capsule Sealing Rate: Monitor the percentage of capsules sealed correctly to ensure that they meet quality standards.
– Machine Downtime: Track the amount of time the machine is not in operation, including maintenance and repair time.
– Production Output: Measure the number of capsules filled per hour or per shift to evaluate the machine’s efficiency.
Guidelines for Monitoring Machine Performance and Efficiency
To maintain optimal machine performance and efficiency, operators should follow these guidelines:
– Set Performance Targets: Establish and track performance targets for filling accuracy, capsule sealing rate, and production output.
– Monitor Machine Parameters: Continuously monitor machine parameters such as filling weight, capsule sealing, and motor speed.
– Analyze Data: Analyze performance data to identify trends, patterns, and areas for improvement.
– Take Corrective Action: Implement corrective action to address any discrepancies in performance or efficiency identified through data analysis.
Regulatory Compliance and Quality Assurance

Adherence to regulatory guidelines is vital in the capsule manufacturing industry, ensuring the production of safe and effective products. Government agencies such as the Food and Drug Administration (FDA) in the United States and the European Medicines Agency (EMA) in the European Union have implemented Good Manufacturing Practice (GMP) guidelines to regulate the production process. These guidelines are essential for maintaining product quality, ensuring patient safety, and preventing contamination.
In capsule manufacturing, GMP guidelines dictate the standards for facilities, equipment, personnel, and processes to ensure compliance with regulations. The FDA and EMA conduct regular inspections to monitor adherence to these guidelines. Manufacturers who fail to comply with regulations may face severe consequences, including fines, product recalls, and even permanent closure of their facilities.
In this section, we will discuss the importance of regulatory compliance, quality control, and assurance in capsule manufacturing. We will also cover the key documentation necessary for regulatory compliance and methods for ensuring consistent product quality and reproducibility.
Role of Quality Control and Assurance
Quality control is an essential aspect of capsule manufacturing, ensuring that products meet the required standards before they are released to the market. Quality assurance, on the other hand, focuses on designing and implementing processes to ensure consistent product quality. This includes setting standards, testing, and auditing to ensure compliance with regulations and internal quality standards.
Quality control and assurance involve a series of steps, including:
- Setting quality standards: Establishing criteria for evaluating product quality, including specifications, testing methods, and acceptance limits.
- Testing and inspection: Carrying out regular testing and inspection of raw materials, in-process materials, and finished products to ensure they meet quality standards.
- Auditing: Conducting regular audits to ensure compliance with quality standards, GMP guidelines, and regulatory requirements.
- Corrective action: Taking corrective action when quality issues are detected, including rework, rejection, or recall of affected products.
- Continuous improvement: Continuously monitoring and improving processes and systems to maintain and enhance product quality.
The role of quality control and assurance is critical in maintaining product quality, ensuring patient safety, and preventing contamination. Manufacturers that prioritize quality control and assurance are more likely to achieve regulatory compliance and maintain a reputation for producing high-quality products.
Key Documentation Needed for Regulatory Compliance, Capsule filling machine manual
Manufacturers must maintain accurate and up-to-date documentation to demonstrate regulatory compliance. The key documentation required includes:
- Certificates of Analysis (COA): A document that certifies the quality and purity of raw materials, in-process materials, and finished products.
- Certificates of Compliance: A document that certifies compliance with regulatory requirements and GMP guidelines.
- Batch records: Records of every batch of product manufactured, including details of raw materials, processing conditions, and testing results.
- Validation reports: Reports that document the validation of processes, including equipment, cleaning validation, and sanitization.
- Complaint files: Records of customer complaints, including investigations and actions taken to resolve the issue.
Maintaining accurate and up-to-date documentation is essential for demonstrating regulatory compliance and ensuring the quality and safety of products.
Ensuring Consistent Product Quality and Reproducibility
Ensuring consistent product quality and reproducibility requires careful control of variables, including raw materials, processing conditions, and equipment. Manufacturers must also implement quality control and assurance measures, including testing, inspection, and auditing to monitor product quality.
Reproducibility can be achieved by:
- Validating processes: Verifying that processes meet specified requirements, including equipment, cleaning validation, and sanitization.
- Standardizing processes: Establishing standardized processes and procedures to ensure consistency.
- Controlling raw materials: Ensuring that raw materials meet specified quality standards.
- Monitoring and controlling product quality: Regularly testing and inspecting products to ensure they meet quality standards.
Manufacturers that prioritize consistent product quality and reproducibility are more likely to achieve regulatory compliance, maintain a reputation for producing high-quality products, and prevent contamination and defects.
Last Word: Capsule Filling Machine Manual
In conclusion, the capsule filling machine manual serves as a valuable resource for anyone involved in the manufacturing process. By following the guidelines and procedures Artikeld in this manual, readers can ensure safe and efficient operation of the equipment, maintain regulatory compliance, and produce high-quality capsules. As the industry continues to evolve, it is essential to remain up-to-date with the latest developments, and this manual provides a solid foundation for understanding the complex world of capsule filling machines.
FAQ Resource
Q: What are the primary components of a capsule filling machine?
A: The primary components of a capsule filling machine include fillers, sealers, and dosers.
Q: How often should I perform maintenance on the capsule filling machine?
A: Regular maintenance should be performed as recommended by the manufacturer, typically daily, weekly, and monthly.
Q: What are the safety features of a capsule filling machine?
A: Safety features include emergency stop buttons, alarm systems, and personal protective equipment (PPE) requirements.